
Lewis, the American chemist who introduced them. Transition metals (d block of the periodic table) often have more than one oxidation state. Members of the carbon family sometimes gain 4 electrons (-4 charge) but in other situations may lose 4 electrons (+4 charge). The nitrogen family elements gain 3 electrons, so their charge is -3. For example, copper usually has a +1 or +2 valence, while iron typically has a +2 or +3 oxidation state. Metal ions may have other charges or oxidation states. This is also the featured periodic table on the cover of our Periodic Table Journal available at Amazon. It includes element names, symbols, groups, atomic numbers, and atomic masses. It features our favorite color scheme of all the tables we’ve made. And 195 looks pretty close to that atomic mass we have there. Group I ( alkali metals) carry a +1 charge, Group II (alkaline earths) carry a +2, Group VII (halogens) carry -1, and Group VIII ( noble gases) carry a 0 charge. This 118 element periodic table is a 1920×1080 HD wallpaper. So an atom of platinum has a mass number of 195. Platinum is sitting right over here if you can see it. 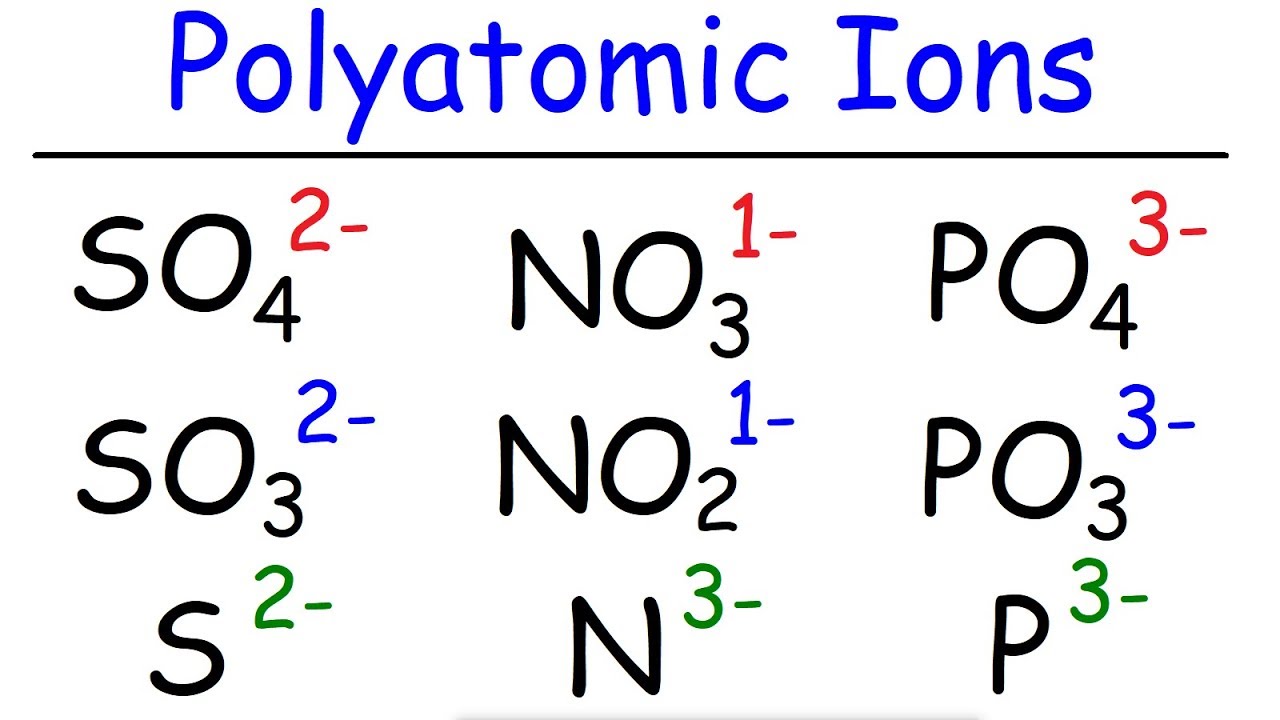
So lets just look up platinum on our periodic table. These diagrams are called Lewis electron dot diagrams, or simply Lewis diagrams, after Gilbert N. They gain two electrons and have a -2 charge. In other words, the atom gains or loses an electron. Here is my way of remembering which charge is with anion and cation: 'A' (in Anion) becomes before 'C' (Cation) in the alphabet. As mentioned above, there are positive and negative ions. For example, the representation for sodium is as follows:Īnd the representation for chlorine is as follows:įor the above diagrams, it does not matter what sides the dots are placed on in Lewis diagrams as long as each side has a maximum of two dots. Ion - unequal amount of protons and electrons in an atom. These dots are arranged to the right and left and above and below the symbol, with no more than two dots on a side. shows the charge of the metal ions in that compound. The figure 3.2.1 below shows how the charge on many ions can be predicted by the location of an element on the periodic table. The number in the name of the compound close compound A substance formed by the chemical union of two or more elements. On the other side of the periodic table, the next-to-last column, the halogens, form ions having a 1 charge. Second, instead of having a circle around the chemical symbol to represent the electron shell, they have up to eight dots around the symbol each dot represents a valence electron. Ions made from alkaline earth metals, the second group on the periodic table, have a 2+ charge. These diagrams have two advantages over the electron shell diagrams. Within a group (family) of elements, atoms form ions of a certain charge.Ĭhemists use simple diagrams to show an atom’s valence electrons and how they transfer. The charge that an atom acquires when it becomes an ion is related to the structure of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
